Monitor every step of the clinical trial process
It’s no secret study managers struggle to select the right technology to connect with patients. With average drop-out rates hovering at 30% across all clinical trials, sites can’t afford to replace inactive participants and put their studies at risk. StudyPages’ intelligent workflow management platform allows teams to connect with participants and monitor real-time insights, so they can stay the course to trial success.
Powerful features at your fingertips
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Suspendisse varius enim in eros elementum tristique. Duis cursus, mi quis viverra ornare, eros dolor interdum nulla, ut commodo diam libero vitae erat.
Reduce manual tracking
Promote team collaboration
Automated communications
Seamless microsite creation
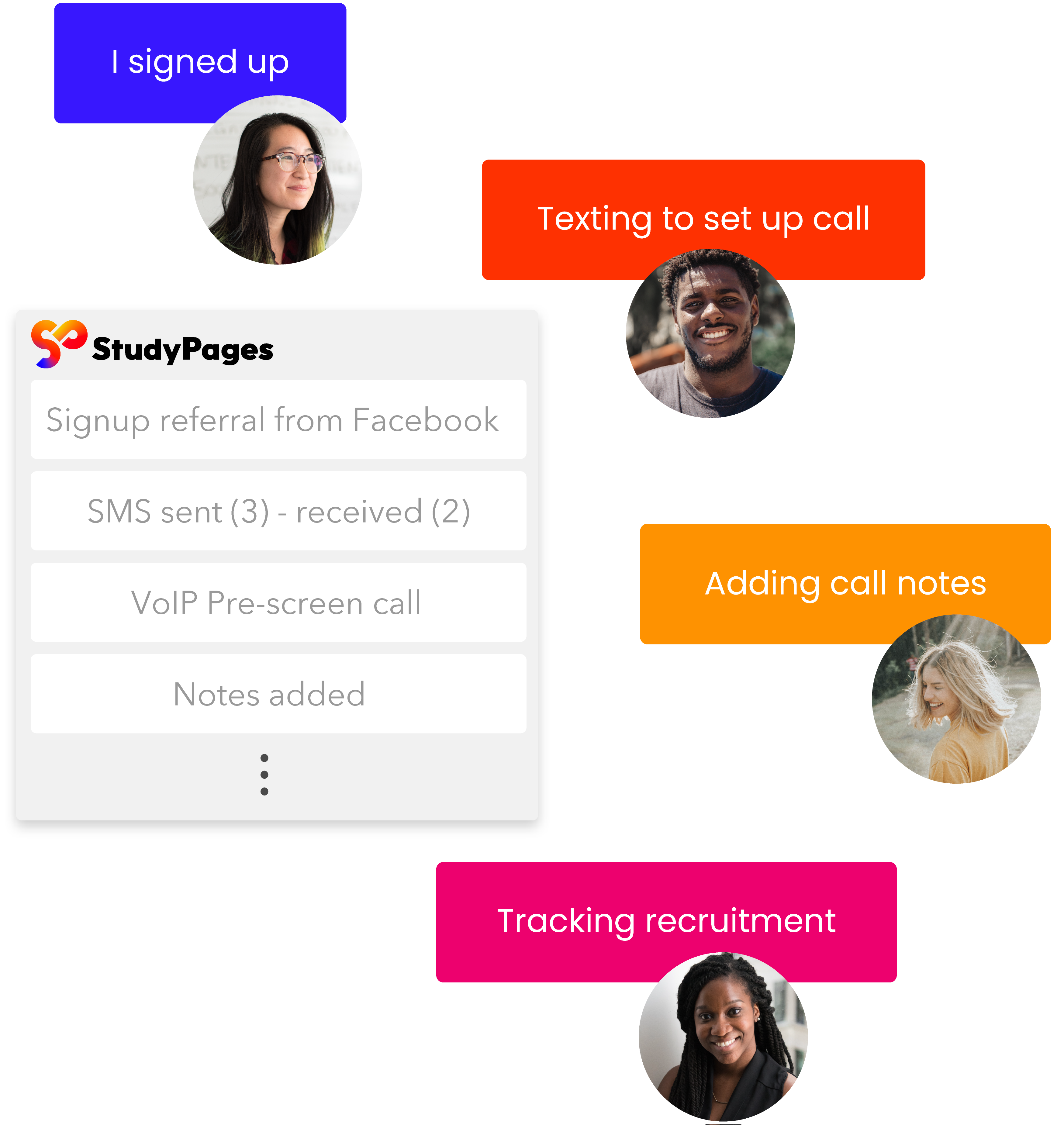
Close trial gaps with collaborative workspaces
Promote operational efficiency with a HIPAA-compliant dedicated workspace. Achieve seamless communication between study teams and leverage automated outreach to engage, track, and retain participants in your pipeline.
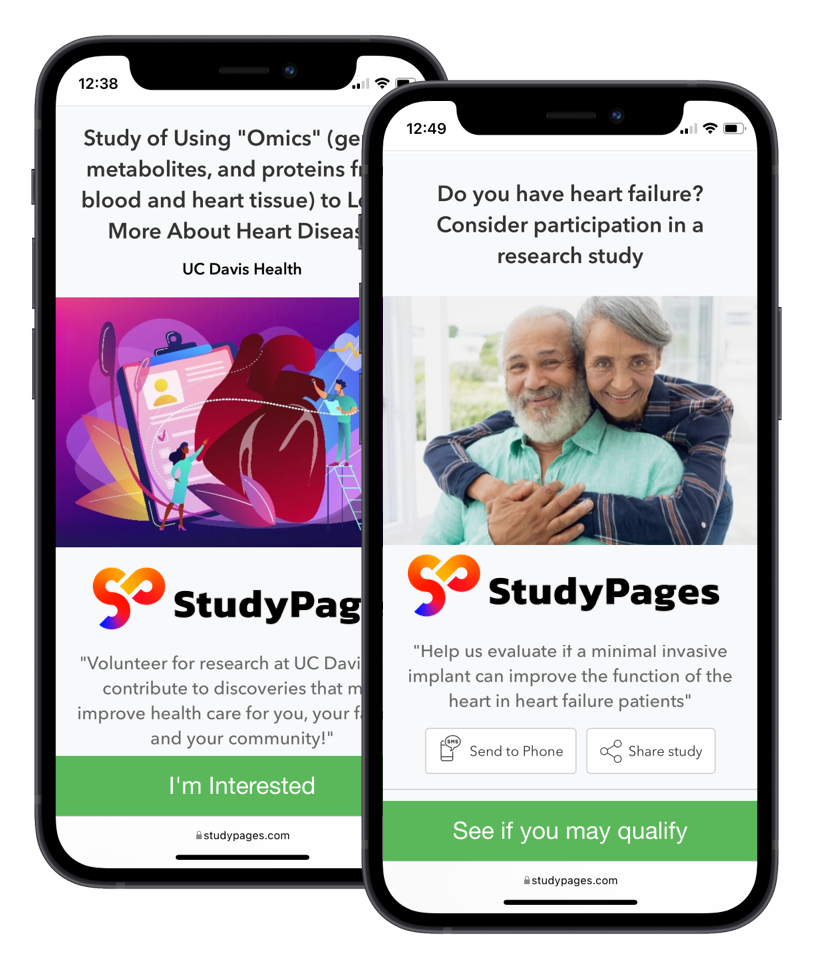
Create branded microsites and study galleries
At StudyPages, we’re breaking down the barriers to trial access by humanizing the clinical discovery process. Build a trustworthy community-facing resource with mobile-friendly landing pages designed to showcase your institution’s research online. Motivate people to enroll in future clinical studies, right on the spot.
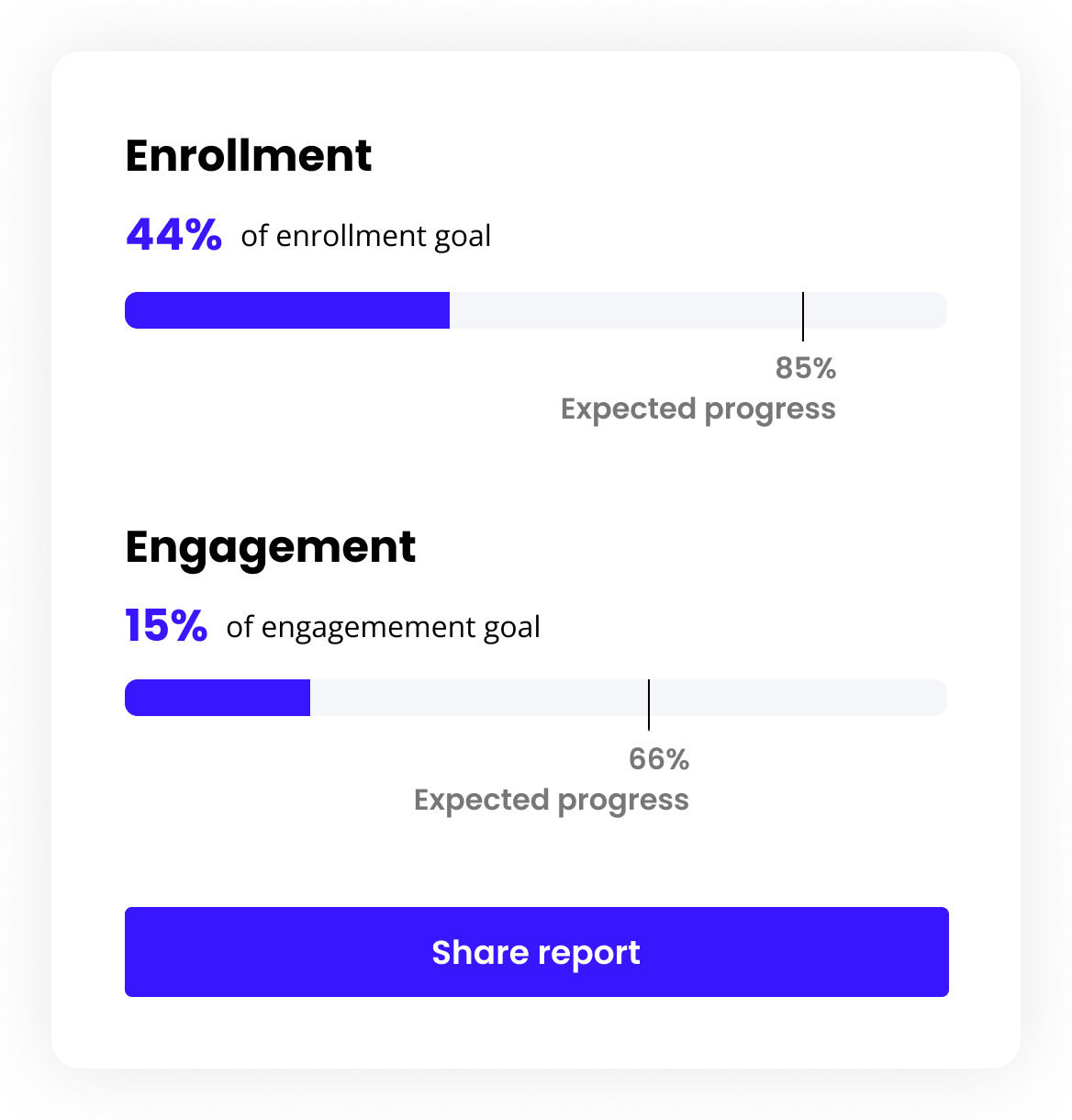
Track and manage recruitment progress
Fix trial bottlenecks by leveraging real-time insights with our intuitive dashboard. View analytics, identify study gaps and challenges, and tackle obstacles head-on, avoiding trial interventions down the line.

Data security & privacy
StudyPages is fully HIPAA and GDPR compliant. We're commited to the protection of our users' privacy. We sell software services and never your data. All information in StudyPages is confidential and securely stored.
Read our data protection and privacy policy
Read our terms of use
Position, Company name
“StudyPages has been a great improvement. We’ve definitely received more referrals coming in through StudyPages, and if not eligible at the time - it starts the conversation, patients are now more informed about what we have to offer and are encouraged to reach back out if still interested in trial participation in the future.”
